TruUCAR is our proprietary technology platform designed for generating high-quality allogeneic CAR-T therapies that can be administered "off-the-shelf". Unlike autologous CAR-T therapies, these products use T cells from non-HLA-matched healthy donors, making them readily available to treat cancer patients, including those who are less suitable for, or have relapsed after, autologous CAR-T cell therapy as well as those with rapidly progressing cancer.
Employing Gene Editing to Reduce the Risk of Patient Rejection
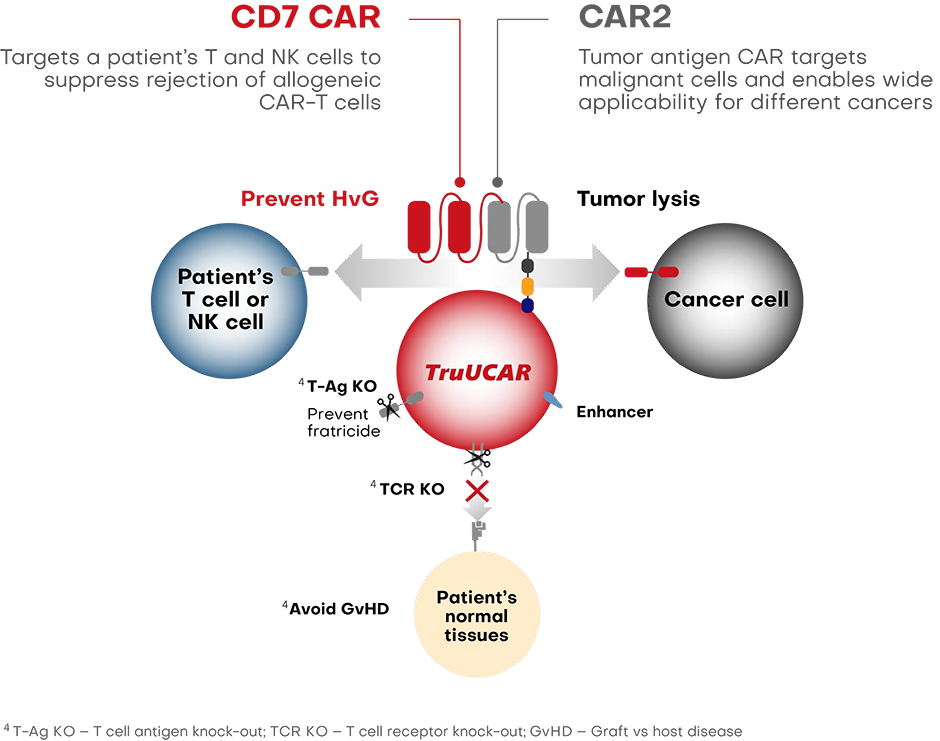
To address the challenge of host-versus-graft rejection (HvG), we are engineering T cells to express a CAR that specifically targets a patient's own T and natural killer (NK) cells (alloreactive killer cells) that would otherwise be directed against the foreign, or allogeneic, CAR-T cells, resulting in rejection by the patient without affecting the recovery of other immune cell compartments during treatment. This feature is designed to allow our allogeneic cell therapies to survive in a patient’s immune system for longer.
To reduce the possibility of graft-versus-host disease GvHD from allogeneic T cells, we are exploring the use of CRISPR/Cas9 to disrupt the T cell receptor alpha constant (TRAC) locus to eliminate surface expression of the TCR complex of our TruUCAR product candidates. Furthermore, to eliminate potential fratricide (self-killing of CAR-T cells during the production process), we utilize CRISPR/Cas9 to disrupt CD7, a pan T and NK marker on the CAR-T cells.
Modular Technology Platform
Since TruUCAR is modular, alternative CAR constructs targeted against different antigens can be applied to TruUCAR to achieve similar potential therapeutic effects. For example, the anti-HvG and anti-GvHD functions can be carried out by a dual CAR design or a single CAR design for dual functions.
In the case of a dual CAR design, one CAR serves a "defensive" purpose, targeting the patient's own alloreactive killer T cells and NK cells while the second CAR serves an "attack" purpose, targeting tumor antigen to eradicate tumor cells. In the case of a single CAR design, the CAR carries out dual functions, targeting both alloreactive killer T cells and NK cells, as well as cancer cells. An enhancer molecule is embedded in the basic construct of TruUCAR to enhance proliferation of TruUCAR T cells.